  
In this regard, if it is assumed that three units of fuel (i.e. And that is 7.It is important to remember the more thermodynamically valuable nature of electricity compared with fuel or heat when comparing VCC COPs to thermal COPs. That's also the heat transferred in, divided by the work or the heat transferred at the high temperature, divided by the work in. The temperature difference between high and low. And that's the high temperature provided by the change. So we can Then we ask for the coefficient of performance for the cycle better prime. And we get x two x one, 0.9304 So we're here, actually were very quite a bit closer. 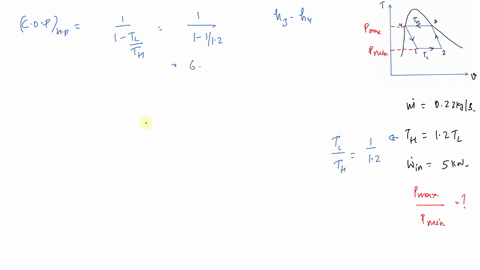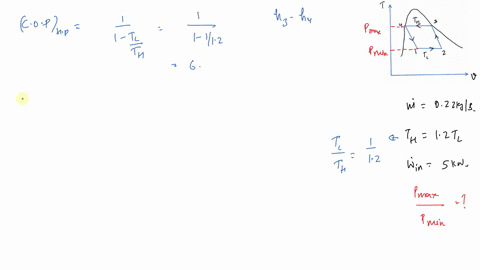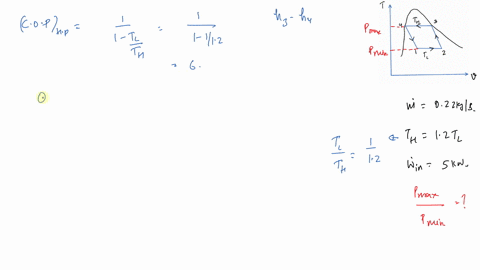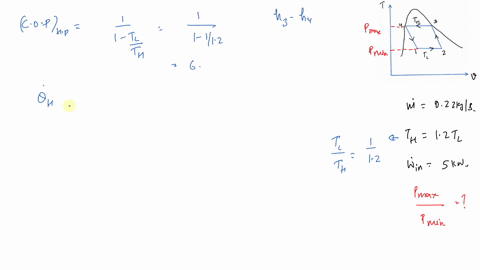
Figure out the, uh, quality factor looking at kind of our end results here on our change from, uh, from liquid to vapor on our entropy in a liquid state. So we can then go to the tables and look up. But now the state one has a temperature of zero degrees C. We know it's the same as it is in ST one. That's 0.8746 killer jewels per kilogram. Over here we have We can look up the entropy for state, too. See what quality factor is given this given this entropy, and that turns out to be a 0.2216 So we're over here. Calvin, Um, then we go down here and figure out for zero degree. And we can look it up for this at the saturated liquid side on that 0.341 7 kg joules per kilogram. So we know that the entropy of four is the same as that of three. And since it's carnal cycle, we have this square or rectangle in our temperature entropy diagram. Um, the heat is transferred so the heat is rejected over this and the heat is transferred in over a zero degree C. Three heat rejection process changes from a saturated paper to accentuated liquids Move from there to there. With our 22 as a working fluid, we know that he did subjected at 40 degrees c. 
I think this probably have Ah, carnal cycle heat pump. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
